Latest Updates
-
 Rich Home Style Masala Chicken Gravy Recipe
Rich Home Style Masala Chicken Gravy Recipe -
 Viral IIT Baba Abhay Singh Secretly Marries Engineer Pratika, Netizens Question His Spiritual Path
Viral IIT Baba Abhay Singh Secretly Marries Engineer Pratika, Netizens Question His Spiritual Path -
 Karishma Tanna and Husband Varun Bangera Share Pregnancy News With Fun Photoshoot, Call Baby ‘Little Miracle’
Karishma Tanna and Husband Varun Bangera Share Pregnancy News With Fun Photoshoot, Call Baby ‘Little Miracle’ -
 Why Women Leaders Are Crucial for Driving Change in India’s Healthcare Sector
Why Women Leaders Are Crucial for Driving Change in India’s Healthcare Sector -
 Who Is Bhoomika Yadav? Meet Raipur’s 19-Year-Old, Youngest Indian Model To Walk For Chanel
Who Is Bhoomika Yadav? Meet Raipur’s 19-Year-Old, Youngest Indian Model To Walk For Chanel -
 Super Crispy Street Style Aloo Tikki Recipe: Your New Favorite Snack!
Super Crispy Street Style Aloo Tikki Recipe: Your New Favorite Snack! -
 Baisakhi 2026: Date, History, Rituals and Cultural Significance Explained
Baisakhi 2026: Date, History, Rituals and Cultural Significance Explained -
 At 83, Amitabh Bachchan Finds Not Working Each Day Disturbing: Why Breaking Routine Triggers Anxiety
At 83, Amitabh Bachchan Finds Not Working Each Day Disturbing: Why Breaking Routine Triggers Anxiety -
 Alia Bhatt Serves Sharp, Subtle Drama in Gaurav Gupta’s Black Three-Piece Tuxedo
Alia Bhatt Serves Sharp, Subtle Drama in Gaurav Gupta’s Black Three-Piece Tuxedo -
 Boss Lady By Zlaata: Workwear That Made Confidence Visible
Boss Lady By Zlaata: Workwear That Made Confidence Visible
Covid-19 Vaccines For Kids: DCGI Grants Emergency Use Nod To 3 Vaccines For Different Age Group Of Children
On Tuesday (26 April), the Drugs Controller General of India (DCGI) approved three Covid-19 vaccines for restricted emergency use authorisation (EUA) in children of various ages.
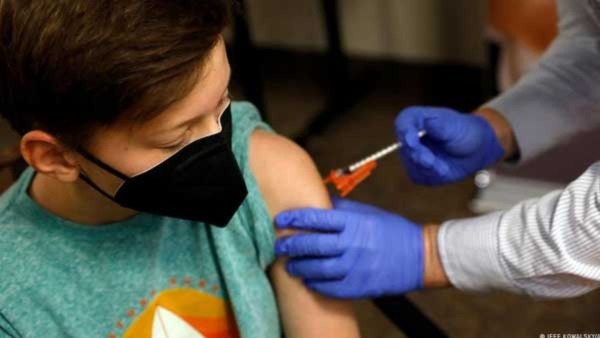
Bharat Biotech's Covaxin received a restricted EUA from the DCGI for children aged 6 to12. Similarly, Biological E's Covid-19 vaccine Corbevax has been approved for children aged 5 to 12 years.
Further, along with Corbevax and Covaxin, the DCGI has also approved Zydus Cadila's Covid-19 vaccine ZyCoV-D for children over the age of 12 years for a two-dose regimen.
This development happened after the Subject Expert Committee's (SEC) discussion of suggestions for the emergency use of Covaxin in children aged 6 to 12 years due to an increase in covid-19 cases in schools.
Covaxin has proven to be effective for adults and is also presently being given to youngsters aged 15 to 18. To give EUA to Covaxin for children aged 6-12 years, experts had requested more information from the corporation, which was delivered on Friday. The SEC was satisfied with the provided data and thus, nods for its EUA among children aged 6-12 years.
As the focus was mainly on the increasing number of cases among children, the SEC has also recommended emergency use of Biological E's Covid-19 vaccine Corbevax and ZyCoV-D.



 Click it and Unblock the Notifications
Click it and Unblock the Notifications












